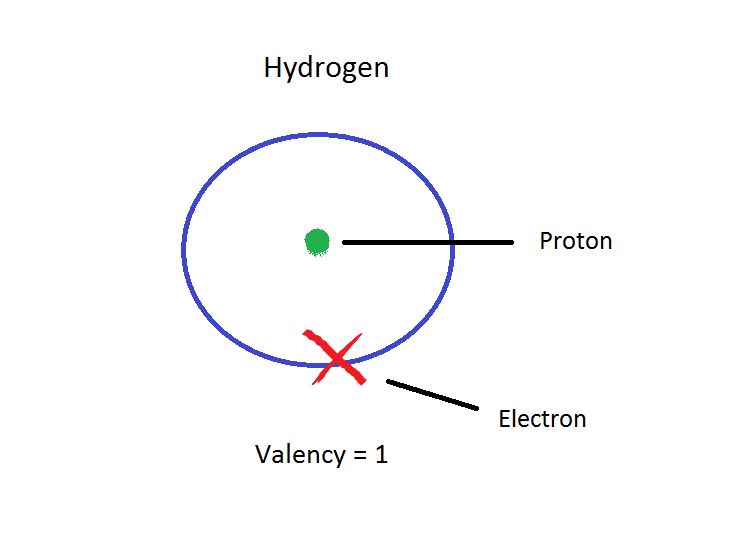
What happens if a compound is actually an ion? A positive or negativeĬharge tells you something about the structure. Try drawing Kekule structures for the following diatomic molecules: For the most part, Kekule structures will be used in This modification of a Lewis structure is sometimesĬalled a Kekule structure. Note that a pair of electrons that forms a bond is usually written as a line Lewis structures are drawings that we use to show how the electrons in a atoms in other rows sometimes have larger numbers of electrons.the Lewis "octet" rule says atoms frequently have 8 valence electrons in.it is common to see atoms with 8 electrons, like neon, in many molecules.most common biological or organic molecules are made from atoms in the.A tally of numbers of electrons in some small The Latin word for eight: second-row elements form bonds so that they can haveĮight valence shell electrons, like neon.įigure IM2.5. Periodic table allows us to think of the Lewis model as "the octet rule", from

Of familiar molecules, takes place mostly in the second row of the The fact that organic and biological chemistry, which constitutes a majority
KRYPTON NUMBER OF VALENCE ELECTRONS PLUS
molecules usually contain even numbers of total electronsįor example, hydrogen, with 1 electron, plus fluorine, with 9 electrons, come.Therefore gain stability if they come together to form an even number overall. Two atoms with odd numbers of electrons may A tally of numbers of electrons in some smallįrom this observation he eventually proposed the notion that electrons are Number (10, 10 and 22, respectively, in these examples).įigure IM2.1. The atoms in almost any known molecular compound, such as water (H 2O),Īmmonia (NH 3) or carbon dioxide (CO 2), you get an even This model still influences the way we think about moleculesįirst of all, Lewis noted that if you count up the number of electrons in all Lewis used some simple observationsĪbout the periodic table to propose an alternate model of how atoms can beīonded together. We know that in most organic compounds, it is common to find twoĬarbon atoms connected together, which is not likely to happen if all carbonsĪre found as charged cations in that case, they would simply repel each other. However, there are some limitations to this way of looking atĬompounds. That were simply pulled together by their opposing charges, like piles of sticky Spheres (such as oxygen anions, O 2-, or carbon cations, C 4+) Small collection of elements, we will need a way to visualize on paper how theseĪt one time, people thought of the atoms in these compounds as charged
In order to think about some of the compounds that can be made with this A model for covalent bonding: Lewis Structures
